|
9/12/2023 0 Comments Na charge periodic table
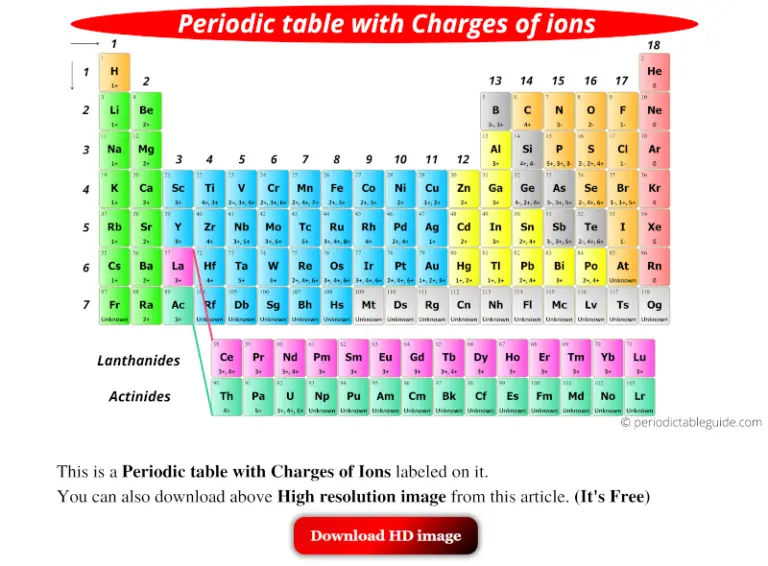
The force of attraction between oppositely charged ions causes them to form an ionic compound. This is because of the basic principle of ‘opposites attract’, where any two objects with opposing electrical charges are attracted towards each other. There is always a force of attraction between two oppositely charged ions. Therefore, the number of negative charges is greater than the number of positive charges and the particle becomes negatively charged overall. If the atom or molecule has gained electrons, the number of electrons is greater than the number of protons. Therefore, the number of positive charges is greater than the number of negative charges and the particle becomes positively charged overall. If the atom or molecule has lost electrons, the number of protons is greater than the number of electrons. Protons are positively charged and electrons are negatively charged. Therefore, the Roman numeral (I) must be used to show the charge of the copper ion.An ion is a charged particle formed when an atom or molecule loses or gains electrons from the outer shells, causing the number of protons and electrons to become unequal.The table below shows that 2 Cu + will balance the charge of 1 S 2.For example, Cu, can have a charge of 1+ or 2+.To find the Roman numeral for the element, first refer to the periodic table. The transition metals below do not need a Roman numeral in the names of their compounds because they only form one ion. /PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
For example, Fe forms Fe 2+ or Fe 3+ ions in compounds. Roman numerals are only used for transition metals that have more than one ion. Transition metals that do not require Roman numerals
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
